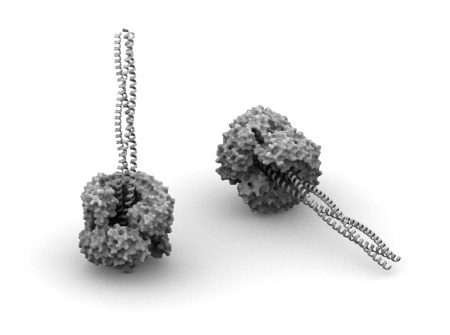
My colleague is at Grenoble on the conference about host-pathogen interactions. Today he sent me important news: Gabriel Waksman (that’s not surprising to anybody interested in structural biology of bacterial pathogenesis) showed structure of usher pore – soon to be published.
Why is that important? Usher is a membrane part of two component system responsible for assembly and transport of fimbriae/pili in gram-negative bacteria – pretty essential element in these organisms. This protein was identified in early 90s (or even earlier) and for quite a while lots of people tried to solve/predict/model its structure. Its structure was assumed to resemble porin – but a large insert right in the middle of beta-barrel gave lots of problems in predicting correct topology. Now we know (at least my colleague saw it, we need to wait) how the final structure looks like and I was also told that its functional aspects have a big relevance to other secretion systems. Have a look on this protein when it’s out (I’ll post definitely about it) – I think you will be surprised even if nuances of host-pathogen interactions are not very appealing to you.
Studying any niche area on the molecular level can be very rewarding. Novel protein fold by itself is not a big deal anymore (it used to be – browse through archives of Nat. Struc. Biol. from several years ago). But putting this novel structure in well known functional context and understanding constrains that led to a new solution is still considered a first-class science.